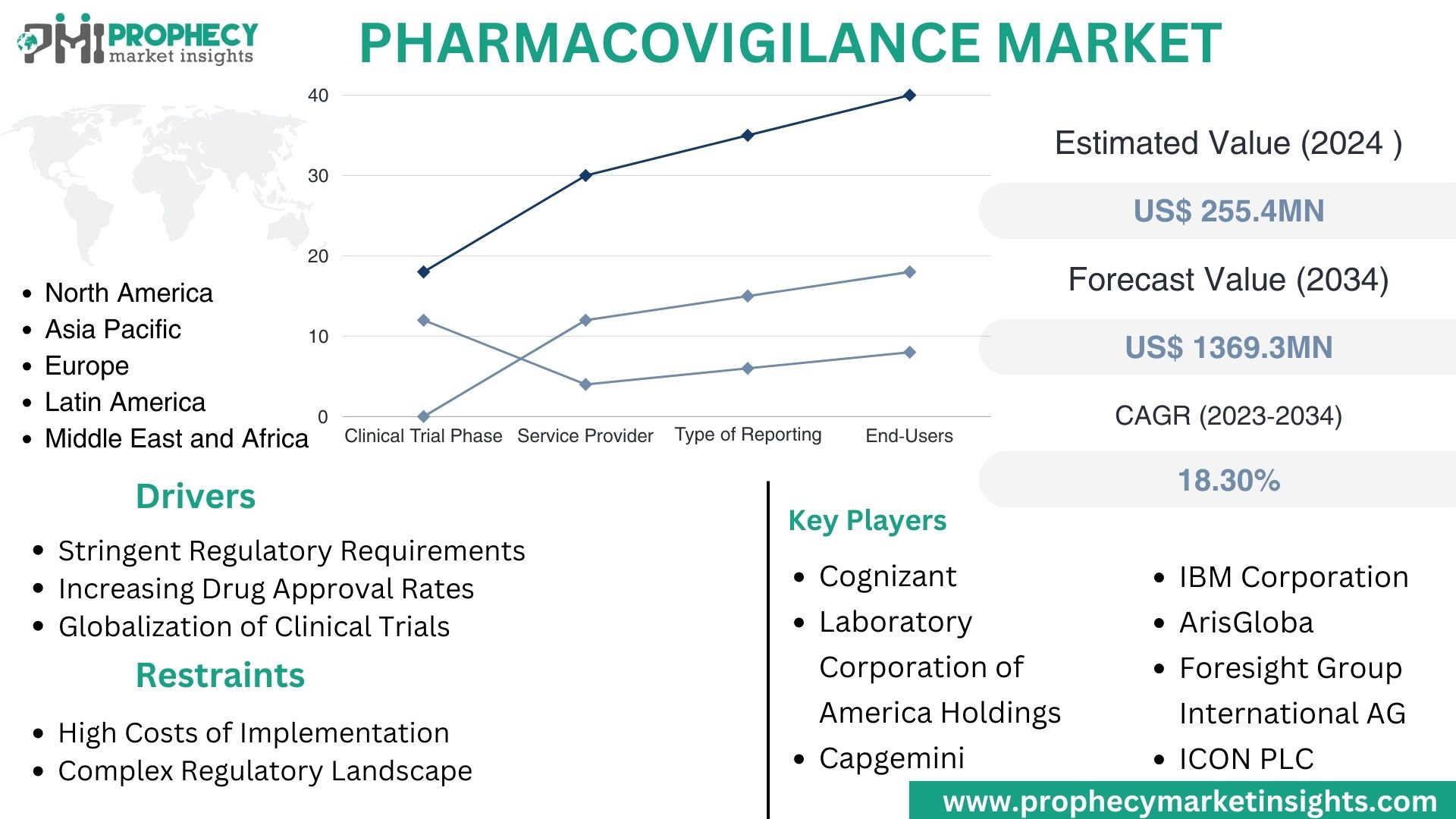
Covina, Feb. 27, 2024 (GLOBE NEWSWIRE) -- “According to the recent research study, the Pharmacovigilance Market size was valued at about USD 8.3 Billion in 2024 and expected to grow at CAGR of 13.50% to extend a value of USD 26.3 Billion by 2034.”
What is Pharmacovigilance?
Market Overview:
Pharmacovigilance is the science and activities related to the detection, assessment, understanding, and prevention of adverse effects or any other drug-related problems. It encompasses the monitoring of the safety of pharmaceutical products before and after they reach the market, aiming to ensure that medicines are used effectively and safely by patients.
The following are important components of pharmacovigilance:
- Adverse Drug Reactions (ADR) Monitoring: Pharmacovigilance involves the systematic collection, analysis, and evaluation of data regarding adverse drug reactions (ADRs) or side effects associated with pharmaceutical products. These reactions may occur during clinical trials or after the drugs are marketed and used by patients.
- Signal Detection and Analysis: Pharmacovigilance activities include the identification of potential safety signals or trends through the analysis of spontaneous reports, clinical trials data, epidemiological studies, literature reviews, and other sources of safety information. Signals may indicate previously unrecognized adverse effects or emerging safety concerns associated with specific drugs.
- Risk Assessment and Management: Pharmacovigilance professionals assess the risks and benefits of pharmaceutical products based on available safety data and scientific evidence. Risk management strategies may involve labeling changes, restrictions on drug use, communication of safety information to healthcare professionals and patients, and post-marketing surveillance programs.
- Regulatory Compliance: Pharmacovigilance is subject to regulatory requirements and guidelines established by health authorities and regulatory agencies worldwide. Pharmaceutical companies are required to establish pharmacovigilance systems, maintain accurate records of safety data, and report adverse events to regulatory authorities in compliance with applicable regulations.
Get Access to Free Sample Research Report with Latest Industry Insights:
https://www.prophecymarketinsights.com/market_insight/Insight/request-sample/4078
*Note: PMI Sample Report includes,
- Overview & introduction of market study
- Revenue and CAGR of market
- Drivers & Restrains factors of market
- Major key players in market
- Regional analysis of the market with a detailed graph
- Detailed segmentation in tabular form of market
- Recent development/news of market
- Opportunities & Challenges of Market
Top Leading Players in Pharmacovigilance Market:
- Accenture
- Cognizant
- Laboratory Corporation of America Holdings
- IBM Corporation
- ArisGloba
- Foresight Group International AG
- ICON PLC.
- Capgemini
- United BioSource Corporation
- Wipro Ltd
Market Dynamics:
Driving Factors:
- Stringent regulatory requirements and guidelines compel pharmaceutical companies to establish robust pharmacovigilance systems and adhere to reporting obligations. Regulatory agencies worldwide mandate the continuous monitoring and reporting of adverse drug reactions (ADRs) to ensure patient safety.
- Increasing awareness of patient safety issues and adverse drug reactions (ADRs) drives the demand for pharmacovigilance activities. Patients and healthcare professionals expect pharmaceutical products to undergo rigorous safety assessment and monitoring to minimize risks and maximize benefits.
- Technological advancements, such as data mining, artificial intelligence, and big data analytics, enhance the efficiency and effectiveness of pharmacovigilance activities. Advanced tools and algorithms enable the timely detection, analysis, and evaluation of safety signals and adverse events.
- The globalization of pharmaceutical markets necessitates harmonization of pharmacovigilance standards and practices across different regions and countries. International collaboration and information-sharing initiatives facilitate the exchange of safety data and enhance pharmacovigilance efforts on a global scale.
- The increasing complexity of drug development, including the emergence of biologics, gene therapies, and combination therapies, underscores the importance of pharmacovigilance in evaluating safety risks associated with novel therapeutic interventions.
Restrain Factors:
- Data Quality and Completeness
- Resource Constraints:
- Complexity of Safety Data Analysis
Emerging Trends and Opportunities in Pharmacovigilance Market:
- AI and big data analytics are revolutionizing pharmacovigilance by enabling more efficient and accurate analysis of safety data. Machine learning algorithms can sift through vast amounts of structured and unstructured data to identify safety signals, trends, and patterns, enhancing signal detection and risk assessment capabilities.
- Pharmacovigilance is adopting advanced methodologies for signal detection, including disproportionality analysis, Bayesian inference, and data mining techniques. These methods enable early detection of safety signals and facilitate proactive risk management strategies to ensure patient safety.
- Real-world data sources, such as electronic health records, claims databases, and patient registries, offer valuable insights into drug safety and effectiveness in real-world clinical practice. Pharmacovigilance is increasingly leveraging RWE to complement traditional clinical trial data and inform post-marketing safety assessments.
- There is a growing emphasis on patient-centered pharmacovigilance approaches that prioritize patient perspectives, experiences, and preferences in safety monitoring and risk communication. Patient-reported outcomes, social media monitoring, and patient engagement initiatives empower patients to contribute to pharmacovigilance efforts and promote shared decision-making in healthcare.
- Pharmacovigilance is integrating pharmacogenomic data to personalize drug safety assessments and optimize medication management based on individual genetic factors. Pharmacogenomic testing identifies genetic variants that influence drug response and susceptibility to adverse drug reactions, enabling tailored pharmacovigilance strategies and precision medicine approaches.
- International collaboration and harmonization initiatives facilitate the exchange of safety information, harmonize pharmacovigilance standards, and streamline regulatory processes across regions and countries. Collaborative networks, such as the International Conference on Harmonisation (ICH), promote best practices and convergence in pharmacovigilance practices worldwide.
Download PDF Brochure:
https://www.prophecymarketinsights.com/market_insight/Insight/request-pdf/4078
Challenges of Pharmacovigilance Market:
- The complexity of safety data analysis, including the interpretation of large datasets, signal detection algorithms, and statistical methodologies, presents challenges in identifying meaningful safety signals and distinguishing true risks from background noise and confounding factors.
- Regulatory Compliance Burden: Compliance with evolving regulatory requirements and reporting obligations imposes a significant burden on pharmaceutical companies and healthcare organizations. The need to navigate complex regulatory frameworks, submit timely and accurate reports, and address regulatory inquiries may strain pharmacovigilance resources and capabilities.
Detailed Segmentation:
Pharmacovigilance Market, By Clinical Trial Phase:
-
-
- Preclinical
- Phase I
- Phase II
- Phase III
- Phase IV
-
Pharmacovigilance Market, By Service Provider:
-
-
- In-house
- Contract
- Outsourcing
-
Pharmacovigilance Market, By Type of Reporting:
-
-
- Spontaneous Reporting
- Intensified ADR Reporting
- Targeted Spontaneous Reporting
- Cohort Event Monitoring
- EHR Mining
-
Pharmacovigilance Market, By End User:
-
-
- Hospitals
- Pharmaceutical Companies
- Others
-
Pharmacovigilance Market, By Region:
-
-
- North America
-
- U.S.
- Canada
-
- Europe
-
- Germany
- UK
- France
- Russia
- Italy
- Rest of Europe
-
- Asia Pacific
-
- China
- India
- Japan
- South Korea
- Rest of Asia Pacific
-
- Latin America
-
- Brazil
- Mexico
- Rest of Latin America
-
- Middle East & Africa
-
- GCC
- Israel
- South Africa
- Rest of Middle East & Africa
-
- North America
-
Regional Analysis:
Regional insights highlight the diverse market dynamics, regulatory landscapes, and growth drivers shaping the Pharmacovigilance Market across different geographic areas. Understanding regional nuances and market trends is essential for stakeholders to capitalize on emerging opportunities and drive market expansion in the Pharmacovigilance sector.
North America market is estimated to witness the fastest share over the forecast period as pharmacovigilance activities in North America involve the ongoing assessment and management of risks associated with pharmaceutical products. Regulatory agencies collaborate with pharmaceutical companies to evaluate safety signals, conduct risk-benefit assessments, and implement risk management strategies to mitigate potential risks to patient health.
Report scope:
| Attribute | Details |
| Market Size 2024 | US$ 8.3 Billion |
| Projected Market Size 2034 | US$ 26.3 Billion |
| CAGR Growth Rate | 13.50% |
| Base year for estimation | 2023 |
| Forecast period | 2024 – 2034 |
| Market representation | Revenue in USD Billion & CAGR from 2024 to 2034 |
| Market Segmentation | By Clinical Trial Phase - Preclinical, Phase I, Phase II, Phase III, and Phase IV By Service Provider – In-house, Contract, and Outsourcing By Type of Reporting – Spontaneous Reporting, Intensified ADR Reporting, Targeted Spontaneous Reporting, Cohort Event Monitoring, and EHR Mining By End User – Hospitals, Pharmaceutical Companies, and Others |
| Regional scope | North America - U.S., Canada Europe - UK, Germany, Spain, France, Italy, Russia, Rest of Europe Asia Pacific - Japan, India, China, South Korea, Australia, Rest of Asia-Pacific Latin America - Brazil, Mexico, Argentina, Rest of Latin America Middle East & Africa - South Africa, Saudi Arabia, UAE, Rest of Middle East & Africa |
| Report coverage | Revenue forecast, company share, competitive landscape, growth factors, and trends |
Key highlights of the Pharmacovigilance Market:
- The pharmacovigilance market is heavily influenced by regulatory requirements and oversight from health authorities worldwide. Pharmaceutical companies are mandated to establish pharmacovigilance systems, adhere to reporting obligations, and comply with regulatory standards to ensure the safety of pharmaceutical products.
- The globalization of pharmaceutical markets and the harmonization of pharmacovigilance standards facilitate international collaboration, information-sharing, and alignment of regulatory practices. Harmonization initiatives promote consistency in pharmacovigilance regulations, guidelines, and reporting requirements across regions and countries.
- Technological advancements, including artificial intelligence (AI), machine learning, big data analytics, and digital health solutions, are transforming pharmacovigilance practices. Advanced technologies enable the efficient analysis of large volumes of safety data, early detection of safety signals, and proactive risk management strategies.
- The growing complexity of drug development, expanding therapeutic landscapes, and rising concerns about medication safety drive the demand for pharmacovigilance services. Pharmaceutical companies, contract research organizations (CROs), and pharmacovigilance service providers seek expertise in safety monitoring, risk assessment, and regulatory compliance.
- Pharmaceutical companies increasingly outsource pharmacovigilance activities to specialized service providers and CROs to optimize resource allocation, reduce operational costs, and ensure compliance with regulatory requirements. Collaboration among stakeholders, including regulatory agencies, healthcare professionals, and industry partners, fosters knowledge sharing, best practices, and innovation in pharmacovigilance.
- Real-world evidence (RWE) generated from electronic health records, claims databases, patient registries, and other sources complements traditional clinical trial data in pharmacovigilance. RWE enhances safety monitoring, post-market surveillance, and risk assessment by providing insights into drug effectiveness, safety outcomes, and treatment patterns in real-world clinical practice.
Any query or customization before buying:
https://www.prophecymarketinsights.com/market_insight/Insight/request-customization/4078
Explore More Insights:
- In Vitro Diagnostics Market - Trends, Analysis and Forecast till 2034
- Diagnostic Labs Market – Trends, Analysis and Forecast till 2034
- Preimplantation Genetic Testing Market- Trends, Analysis and Forecast till 2034
Blog: www.prophecyjournals.com
Follow us on:
LinkedIn | Twitter | Facebook |YouTube
