Dublin, April 23, 2024 (GLOBE NEWSWIRE) -- The "U.S. Gene Therapy Market Size, Share & Trends Analysis Report by Indication (Multiple Myeloma, Spinal Muscular Atrophy, Inherited Retinal Disease), Route Of Administration, Vector Type (Lentivirus, AAV, Adenovirus), and Segment Forecasts, 2024-2030" report has been added to ResearchAndMarkets.com's offering.
The U.S. gene therapy market size is anticipated to reach USD 10.40 billion by 2030, expanding at a CAGR of 19.21% during 2024-2030
The market growth can be attributed to various factors such as the expanding area of advanced therapies along with gene delivery technologies and increased competition among key players focused on the commercialization of their therapies. 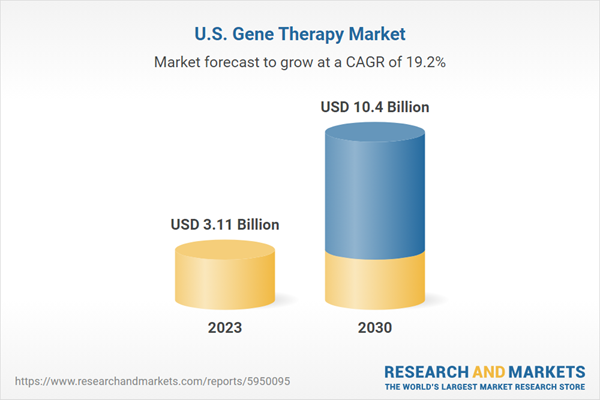
Similar to many other industries, the COVID-19 pandemic has had an impact on the U.S. market for gene therapy. While the market is expected to grow significantly in the coming years, the pandemic has caused delays in clinical trials and disruptions in the supply chain. The pandemic also highlighted the importance of gene therapy in treating and preventing diseases, which could lead to increased funding and investment in the field. Overall, while the pandemic has caused some challenges for the U.S. gene therapy industry, there is still significant potential for growth and advancement in the coming years.
The gene therapy pipeline in the U.S. is currently quite robust, with several promising therapies in various stages of development. Many of these therapies focus on rare genetic diseases, such as spinal muscular atrophy, choroideremia, Duchenne muscular dystrophy, and hemophilia. In addition, gene editing technologies like CRISPR-Cas9 are being used to develop therapies that can target specific genetic mutations with greater precision.
For instance, in December 2023, Vertex Pharmaceuticals Incorporated and CRISPR Therapeutics jointly made the announcement of CASGEVY (exagamglogene autotemcel [exa-cel]), a genome modified cell therapy using CRISPR/Cas9 technology, has been approved by the U.S. FDA for treating sickle cell disease in patients aged 12 and above who have recurrent vaso-occlusive crises (VOCs).
However, drug companies have attached very high prices to these narrowly targeted treatments, resulting in one being pulled from the market in Europe and another struggling to attract patients. For instance, in November 2023, Sarepta Therapeutics revealed the price of the drug per infusion of the Elevidys, gene therapy for Duchenne muscular dystrophy (DMD), which is approximately USD 3.2 million.
U.S. Gene Therapy Market Report Highlights
- Large B-cell lymphoma dominated the indication segment with the largest revenue share of 36.04% in 2023. This is attributed to the high prevalence of this type of cancer in the country. Additionally, gene therapy has shown promising results in treating this disease, leading to increased adoption by healthcare providers and patients.
- The intravenous route of administration held the larger market share in 2023 and is expected to grow at a higher CAGR during the forecast period. This route of administration involves the use of viruses, such as adenovirus, retrovirus, or lentivirus, to deliver the desired gene into the patient's cells.
- Based on vector type, the lentivirus segment dominated with the largest revenue share in 2023. On the other hand, AAV is anticipated to grow at the highest CAGR from 2024 to 2030, owing to the rising demand and their usage in clinical trials due to the higher accuracy level in delivering the gene to the region of interest.
Companies Featured
- Amgen Inc.
- Novartis AG
- F. Hoffmann-La Roche
- Gilead Sciences, Inc.
- bluebird bio, Inc.
- Bristol-Myers Squibb Company
- Legend Biotech.
- BioMarin.
- uniQure N.V.
- Merck & Co.
- Sarepta Therapeutics, Inc.
- Krystal Biotech, Inc.
- CRISPR Therapeutics.
Key Attributes:
| Report Attribute | Details |
| No. of Pages | 90 |
| Forecast Period | 2023 - 2030 |
| Estimated Market Value (USD) in 2023 | $3.11 Billion |
| Forecasted Market Value (USD) by 2030 | $10.4 Billion |
| Compound Annual Growth Rate | 19.2% |
| Regions Covered | United States |
Key Topics Covered:
Chapter 1. Methodology and Scope
Chapter 2. Executive Summary
2.1. Market Outlook
2.2. Segment Snapshot
2.3. Competitive Landscape Snapshot
Chapter 3. Market Variables, Trends, & Scope
3.1. Market Lineage Outlook
3.1.1. Parent Market Outlook
3.1.2. Related/Ancillary Market Outlook
3.2. Market Dynamics
3.2.1. Market Driver Analysis
3.2.1.1. Robust gene therapy pipeline
3.2.1.2. Introduction of technological advancements
3.2.1.3. Growing prevalence of target diseases
3.2.1.4. Increasing investments from companies and partnerships
3.2.2. Market Restraint Analysis
3.2.2.1. High prices of gene therapy
3.2.2.2. Limited manufacturing capacity
3.3. Industry Analysis Tools
3.3.1. Porter's Five Forces Analysis
3.3.2. PESTEL Analysis
3.3.3. COVID-19 Impact Analysis
Chapter 4. Indication Business Analysis
4.1. Indication Segment Dashboard
4.2. U.S. Gene Therapy Market Indication Movement Analysis
4.3. U.S. Gene Therapy Market Size & Trend Analysis, by Indication, 2018 to 2030 (USD Million)
4.4. Large B-Cell Lymphoma
4.4.1. Large B-cell lymphoma market estimates and forecasts, 2018 - 2030 (USD Million)
4.5. Multiple Myeloma
4.6. Spinal Muscular Atrophy (SMA)
4.7. Acute Lymphoblastic Leukemia (ALL)
4.8. Melanoma (Lesions)
4.9. Inherited Retinal Disease
4.10. Beta-Thalassemia Major/SCD
Chapter 5. Route of Administration Business Analysis
5.1. Route of Administration Segment Dashboard
5.2. U.S. Gene Therapy Market Route of Administration Movement Analysis
5.3. U.S. Gene Therapy Market Size & Trend Analysis, by Route of Administration, 2018 to 2030 (USD Million)
5.4. Intravenous
5.5. Others
Chapter 6. Vector Type Business Analysis
6.1. Vector Type Segment Dashboard
6.2. U.S. Gene Therapy Market Vector Type Movement Analysis
6.3. U.S. Gene Therapy Market Size & Trend Analysis, by Vector Type, 2018 to 2030 (USD Million)
6.4. Lentivirus
6.5. RetroVirus & gamma RetroVirus
6.6. AAV
6.7. Modified Herpes Simplex Virus
6.8. Adenovirus
Chapter 7. Competitive Landscape
7.1. Company Categorization
7.2. Strategy Mapping
7.3. Company Market Position Analysis, 2023
7.4. Company Profiles/Listing
7.4.1. Overview
7.4.2. Financial Performance
7.4.3. Product Benchmarking
7.4.4. Strategic Initiatives
For more information about this report visit https://www.researchandmarkets.com/r/jkn955
About ResearchAndMarkets.com
ResearchAndMarkets.com is the world's leading source for international market research reports and market data. We provide you with the latest data on international and regional markets, key industries, the top companies, new products and the latest trends.
Attachment
