Bionic Vision Technologies Announces Interim Pilot Study Results of the BVT Bionic Eye System Designed to Help the Blind Achieve Greater Mobility and Independence
13 janv. 2020 11h45 HE
|
Bionic Vision Technologies
SAN FRANCISCO, Jan. 13, 2020 (GLOBE NEWSWIRE) -- Bionic Vision Technologies today announced the interim results of a pilot study involving four patients with late-stage Retinitis Pigmentosa (RP)...
ProQR Announces First Patient Dosed in Phase 1/2 Aurora Trial of QR-1123 for Autosomal Dominant Retinitis Pigmentosa
11 déc. 2019 07h00 HE
|
ProQR Therapeutics N.V.
LEIDEN, Netherlands and CAMBRIDGE, Mass., Dec. 11, 2019 (GLOBE NEWSWIRE) -- ProQR Therapeutics N.V. (Nasdaq:PRQR), a company dedicated to changing lives through the creation of transformative RNA...
ProQR Receives Orphan Drug Designation from FDA for QR-1123 for Autosomal Dominant Retinitis Pigmentosa
21 nov. 2019 07h00 HE
|
ProQR Therapeutics N.V.
LEIDEN, Netherlands and CAMBRIDGE, Mass., Nov. 21, 2019 (GLOBE NEWSWIRE) -- ProQR Therapeutics N.V. (Nasdaq:PRQR), a company dedicated to changing lives through the creation of...
ProQR Receives Fast Track Designation from FDA for QR-1123 for Autosomal Dominant Retinitis Pigmentosa
09 sept. 2019 07h00 HE
|
ProQR Therapeutics N.V.
LEIDEN, Netherlands and CAMBRIDGE, Mass., Sept. 09, 2019 (GLOBE NEWSWIRE) -- ProQR Therapeutics N.V. (Nasdaq:PRQR), a company dedicated to changing lives through the creation of...
First Clinical Program Initiated Using Nanoparticles to Treat Retinitis Pigmentosa
23 avr. 2019 11h00 HE
|
2Ctech, Inc.
IRVINE, Calif., April 23, 2019 (GLOBE NEWSWIRE) -- 2Ctech, Inc., a privately-held development stage company focused on the application of nanoparticle technologies for the treatment of retinal...
ProQR Doses First Patient in Phase 1/2 STELLAR Trial of QR-421a for Usher Syndrome Type 2
11 mars 2019 08h00 HE
|
ProQR Therapeutics N.V.
LEIDEN, Netherlands and CAMBRIDGE, Mass., March 11, 2019 (GLOBE NEWSWIRE) -- ProQR Therapeutics N.V. (Nasdaq:PRQR), a company dedicated to changing lives through the creation of transformative RNA...
ProQR In-licenses Worldwide Rights to Ophthalmology Drug Candidate from Ionis Pharmaceuticals
29 oct. 2018 07h33 HE
|
ProQR Therapeutics N.V.
ProQR receives exclusive worldwide license for IONIS-RHO-2.5Rx, now QR-1123, for autosomal dominant retinitis pigmentosa (adRP), a rare inherited form of blindness with no approved therapy A first in...
Drug Candidates in Retinitis Pigmentosa Therapeutics Pipeline to Develop More as Gene Therapy: P&S Market Research
27 mars 2018 05h39 HE
|
P&S Market Research
NEW YORK, March 27, 2018 (GLOBE NEWSWIRE) -- According to a new research report titled “Retinitis Pigmentosa Therapeutics Pipeline Analysis, 2017” published by P&S Market Research, Retinitis...
GenSight Biologics Enrolls First Patient in Pivotal Phase III Program of GS010 in Leber’s Hereditary Optic Neuropathy
29 févr. 2016 07h00 HE
|
GenSight Biologics
PARIS, Feb. 29, 2016 (GLOBE NEWSWIRE) -- GenSight Biologics S.A. (GenSight), a clinical-stage biotechnology company discovering and developing novel gene therapies for neurodegenerative retinal...
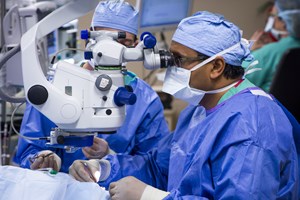
UCHealth Eye Center Restores Limited Sight to Blind Patient with Region's First Bionic Eye Surgery
10 déc. 2015 09h10 HE
|
UCHealth
AURORA, Colo., Dec. 10, 2015 (GLOBE NEWSWIRE) -- The UCHealth Eye Center at University of Colorado Hospital has performed the first "bionic eye" procedure in the Rocky Mountain region, restoring...